Impact of PHI XPS instruments on scientific discoveries
Publishing recent discoveries in science and technology in peer-reviewed literature is a critical function of members of the research community. It enables us to recognize the latest progress and inspires further advances. We are looking back onto the year 2021 – on the impact Physical Electronics XPS instruments, such as PHI VersaProbe, PHI Quantera, and PHI Quantes, have had in supporting scientific breakthroughs.
Over 4500 scholar publications, including peer-reviewed articles and book chapters, have been published in 2021 using PHI XPS instruments. There were more than 60 papers published in the high-impact journals (Nature and Science group), with more than 30% studying novel energy-related materials, such as batteries, fuel cells, and solar cells, and 20%. on nanomaterials.
PHI XPS instruments were used to study a large range of materials of high technological and research importance - surface treatment for inactivating SARS-CoV‑2 Virus1, oxygen evolution electrocatalysts2,3, CO2 reduction catalysts4, single-atom catalysts5,6, graphene-based materials for electrochemical materials7, ion-selective biological channels8, perovskite solar cells9,10, novel 2D materials – MXenes11 and magnetene12, novel magnetic materials based on perovskites for cooling applications13, organic multilayered semiconductors14, next-generation cathodes for lithium batteries15,16, 17, sodium-ion batteries18, lithium-ion batteries19 and solid-state batteries20.
One paper with the highest number of citations (51 citations in less than one year) published in ACS Nano is from our customers at Drexel University studying novel types of 2D materials - highly conductive Ti3C2 MXene.11 “MXenes have been utilized in various fields, including energy storage and conversion, electromagnetic interference shielding, nanocomposites, sensors, and biomedical applications. MXenes have several significant advantages over graphene and many other conducting nanomaterials: MXenes form stable colloidal solutions without additives or surfactants, and they can easily be processed using the cheapest and safest solvent−water”. Using the PHI VersaProbe Scanning XPS Mircoprobe instrument, Mathis and co-workers demonstrated close to perfect stoichiometry and elimination of oxygen from the carbon sublattice, which may have contributed to the improved oxidation stability of the Al−Ti3C2.
XPS is becoming one of the main characterization techniques scientists use in their discovery of novel battery materials.
In a collaborative work led by the Massachusetts Institute of Technology (published in Nature Energy and cited 16 times in less than a year17), a novel sulfonamide-based electrolyte was shown to enable stable ultra-high-voltage cycling of a high-nickel cathode in practical Li metal batteries. To characterize the cathode-electrolyte interface, XPS measurements were conducted on the surface of the cathodes after the 100 cycles. Authors compared C 1s, and F 1s spectra from a cathode cycled in the novel electrolyte to that in a reference and discovered more LiF-like inorganic components and less organic components pointing to the less reactive and corrosive nature of the novel electrolyte.
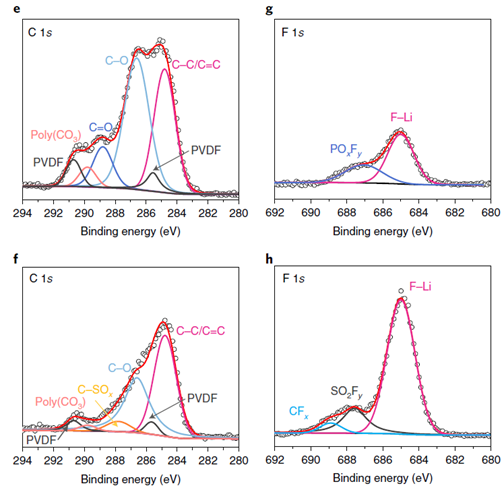 Fig. 1 XPS analysis for the cathodes cycled in the novel sulfonamide-based electrolyte (f and h) and reference (e and g) electrolytes for 100 cycles. PVDF= polyvinylidene fluoride
Fig. 1 XPS analysis for the cathodes cycled in the novel sulfonamide-based electrolyte (f and h) and reference (e and g) electrolytes for 100 cycles. PVDF= polyvinylidene fluoride
Another important collaborative work between researchers at Zhengzhou University, Penn State University, and the University of Texas at Austin (published in Nature communication and cited nine times in less than a year21) focuses on lithium-sulfur (Li–S) batteries that couple a Li anode with a sulfur (S) cathode. Authors address two significant problems in Li-S batteries - interfacial instability of the lithium-metal anode and polysulfide migration from the cathode - by using a promising electrolyte additive BTT, which involves an exchange reaction on the Li-metal anode and electrochemical polymerization on the sulfur cathode.
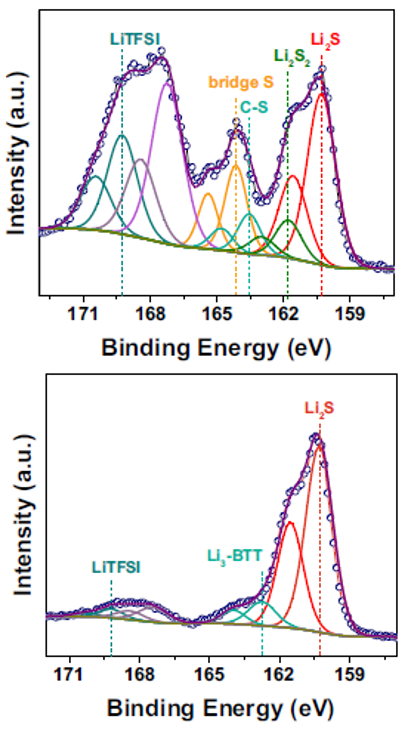 Fig. 2 XPS data for the recharged cathode after 5 cycles at 0.05C rate for the BTT cell (top), and the discharged cathode after 5 cycles for BTT cell (bottom)
Fig. 2 XPS data for the recharged cathode after 5 cycles at 0.05C rate for the BTT cell (top), and the discharged cathode after 5 cycles for BTT cell (bottom)
They used the PHI VersaProbe instrument for depth profile characterization of the dual solid-electrolyte interface in asymmetric Li cell after cycling. The depth profile provided information on the chemical environment between Li and the S-H functional group of BTT based on the Li, S, and F spectra. The homogeneity of Li3-BTT and the apparent change of Li-F demonstrated that the BTT SEI is composed of organic lithium and inorganic lithium compounds. “Compared with the BTT electrode, the SEI formed in the commercial electrolyte is fragile and has poor ionic conductivity, with uneven composition. It also includes more reduced products, such as SO32- and S2- (from LiTFSI) in the S 2p spectrum and side reaction products of LiOH (55.0 eV in Li 1s spectrum), Li–O, and Li–F in the Li 1s spectrum.”
XPS was also collected to further understand the chemical composition of the charged product. The S 2p peaks of the bridged S (S–S) and the S bonded to the phenyl ring (C-S) are found at 164.1 eV and 163.5 eV in the recharged product, respectively. The presence of Li2S and Li2S2 are due to the incomplete charging of the cell.
We are very proud to be a part of the exciting discoveries our customers make. Read more about them in the papers cited below.
- https://doi.org/10.1021/acsami.1c15505
- https://doi.org/10.1038/s41560-021-00925-3
- https://doi.org/10.1002/adfm.202106229
- https://doi.org/10.1038/s41467-021-25573-9
- https://www.science.org/doi/10.1126/sciadv.abd9210
- https://doi.org/10.1038/s41557-021-00734-x
- https://doi.org/10.1038/s41598-021-01154-0
- https://doi.org/10.1021/acsnano.1c07210
- https://doi.org/10.1038/s41467-021-26754-2
- https://doi.org/10.1038/s41565-021-01010-2
- https://doi.org/10.1021/acsnano.0c08357
- https://www.science.org/doi/10.1126/sciadv.abk2041
- https://doi.org/10.1038/s41598-021-99755-2
- https://doi.org/10.1002/smtd.202001264
- https://doi.org/10.1021/acsami.1c15271
- https://doi.org/10.1038/s41467-021-26815-6
- https://doi.org/10.1038/s41560-021-00792-y
- https://doi.org/10.1002/adfm.202109694
- https://doi.org/10.1038/s41467-021-26073-6
- 10.33774/chemrxiv-2021-4dnn0
- https://doi.org/10.1038/s41467-021-23155-3



